I would like to thank the authors for their wonderful contributions to this book and I believe this will significantly improve our understanding of the role of oxidative stress in the pathology and pathophysiology of cardiovascular disease and other related human conditions.
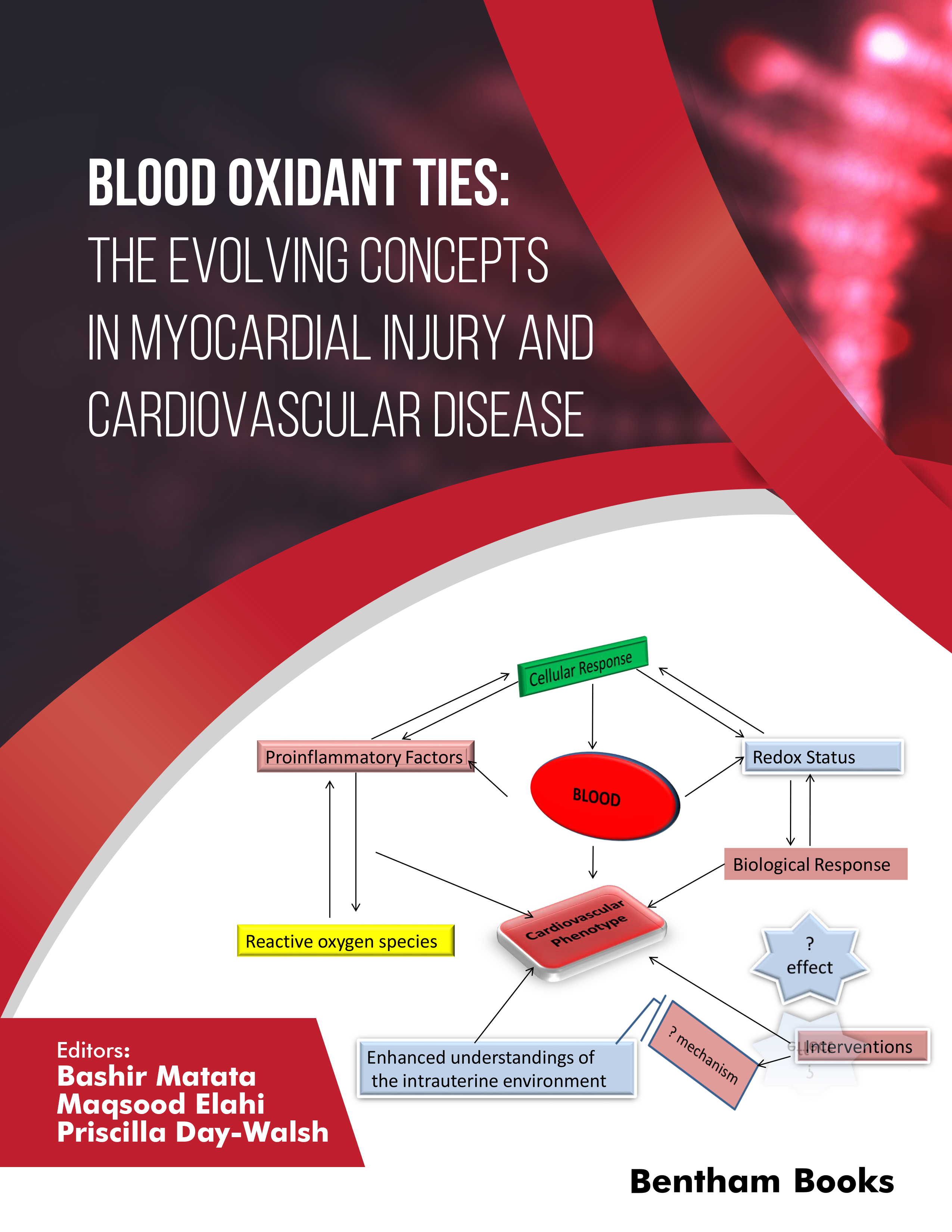
Oxidative stress is described as an intracellular and extracellular imbalance between the production of free radicals and the availability of appropriate antioxidant species and the effectiveness of these molecules to counter these species. Evidence from the literature suggests that oxidative stress is a major contributor to the pathogenesis and pathophysiology of cardiovascular disease.
A major contributor to oxidative stress is a family of reactive oxygen species (ROS) that are unstable molecules and contain one or more unpaired electrons in atomic or molecular orbitals that readily react with other molecules. The widespread production of ROS damages the plasma membrane and stimulates the release of various proinflammatory agents. Several proteins become denatured; for example, receptors, ionic channels, transporters, or components of transduction pathways through oxidation by ROS. Altered protein structure inhibits their functions leading to the disruption of vital cellular processes. Not only can ROS contribute to oxidative stress in general, but individual species of ROS also have their own distinct properties and may activate diverse signalling pathways. Activation of these signalling pathways leads to distinct pathological changes associated with cardiovascular disease cascades. For example, the onset of reperfusion injury is further exacerbated by the activation and infiltration of the infarcted area by polymorphonuclear leukocytes (PMNs). Several studies have identified the release of different leukocyte intracellular factors during PMN activation, such as selectins and b2-integrins to be related to the magnitude of tissue damage.
Oxidative stress is a major contributor to ischaemia reperfusion injury-mediated myocardial infarction. The onset of coronary ischemia deprives the heart muscles of nutrients and oxygen in the areas distal to the site of occlusion, rendering cardiomyocytes unable to undergo aerobic metabolism to support their energy requirements. Homeostatic intracellular signalling systems, such as the hypoxia-inducible factor (HIF) transcription factor cascade sense the low oxygen environment. This in turn stimulates the upregulation of numerous compensatory mechanisms which are ultimately involved in elevating anaerobic glycolysis and promoting angiogenesis and vascularization. The increased anaerobic metabolism increases the production of lactic acid hence metabolic acidosis. This leads to myocyte death and the expansion of the size of the original area of the infarct. Under normal aerobic conditions, the myocardium generally metabolises relatively high levels of adenosine triphosphates (ATP). In contrast, during ischemia, the shift in energy production to glycolysis results in the inefficient production of ATP and constitutes a pathological feature, and if not reversed, early may lead to complications such as heart failure and ischemia-induced atrial or ventricular fibrillation. Despite the widespread use of fibrinolytic agents and new types of angioplasty procedures for the treatment of myocardial infarction, often new sets of complications persist. These include the occurrence of extensive tissue injury caused by myocardial reperfusion through the reintroduction of oxygen to previously ischemic tissues because of the excessive generation of reactive oxygen species (ROSs) and depletion of antioxidants.
Interestingly, more recent evidence has shown that the activation of proinflammatory cells in particular PMN, is closely linked to the activation of other cells involved in the inflammatory response. For example, during myocardial ischemia-reperfusion injury, it has been shown that the activity of neutrophils is also modulated by lymphocytes and macrophages.
Myocardial reperfusion injury (MIR) accounts for about half of the final damage caused by cardiac ischemia-reperfusion. Considering the importance of clinical models involving cardiac ischemia-reperfusion, such as acute myocardial infarction (AMI) and cardiac surgery (CS), many therapies have been developed to prevent MIR. However, despite encouraging results in experimental studies in this innovation journey, no therapy has shown substantial clinical benefits. The view of the therapeutic role of antioxidants as one of the potential treatments to prevent MIR in these settings arises from the known burst of reactive oxygen species occurring from the onset of myocardial reperfusion. Although antioxidants have shown some clinical benefit in the prevention of reperfusion arrhythmias in CS, no antioxidant alone has successfully reduced the final infarct size or caused improvement in other long-term clinical outcomes. Therefore, recently novel strategies have been developed that combine nanotechnology as a mode of delivery of antioxidant therapies that have shown to result in better clinical benefits throughout the innovation journey.
This book provides an in-depth analysis of the relationship between oxidative stress and cardiovascular as follows:
1. The current knowledge of the biological concept of oxidative stress and its role in the pathogenesis of the cardiovascular disease.
2. The interaction between oxidative stress, activation of different leukocytes, and the release of factors involved in the generation of myocardial ischaemia reperfusion (MIR) injury.
3. The interaction between oxidative stress and lipid metabolism i.e. metabolomics as primary drivers for myocardial injury and cardiovascular disease.
4. The interaction between oxidative stress and in-utero programming of the cardiovascular disease.
5. The role of antioxidants in the prevention of cardiovascular disease and acute myocardial infarction.
6. An update on the stage of development of new nanotechnologies-based antioxidants therapies for the prevention of myocardial injury.
The authors are collectively known for their extensive contributions in the field of oxidative stress and cardiovascular disease research and would like to dedicate this book to the support of different funding organisations that have contributed to their work over the years. In addition, the authors acknowledge and wish to thank work colleagues, friends, and families collectively for their support especially when balancing daily responsibilities and finding sufficient time to write their contributions to this magnificent book in the middle of the covid-19 pandemic.
We welcome reader's suggestions and comments for future improvements.
Bashir Matata
Central Liverpool Primary Care Hub
81 London Road
Liverpool, L3 8JA
United Kingdom